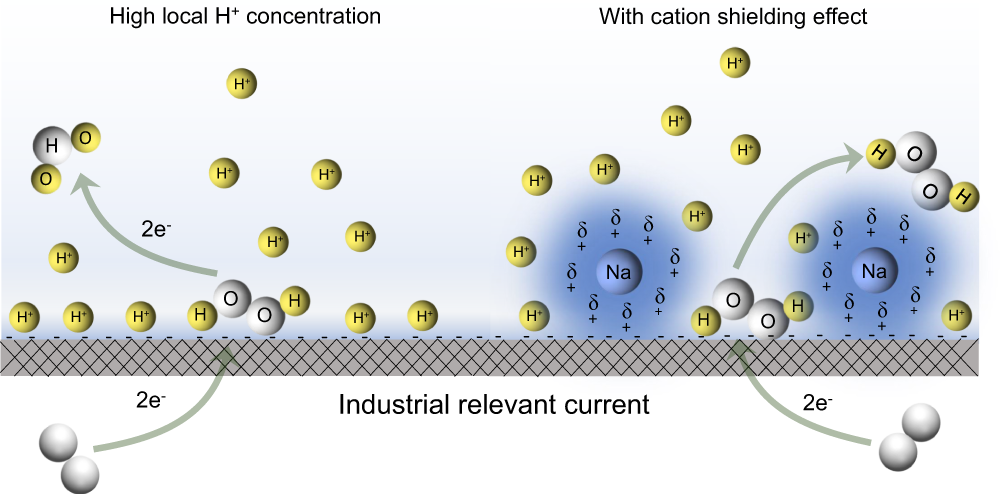
Electrochemical oxygen reduction to hydrogen peroxide at practical rates in strong acidic media | Nature Communications
![Amazon.com: Very High Level Hydrogen Peroxide H2O2 Test Strips, 2-10%, Time Based Test [Vial of 50 Strips] : Industrial & Scientific Amazon.com: Very High Level Hydrogen Peroxide H2O2 Test Strips, 2-10%, Time Based Test [Vial of 50 Strips] : Industrial & Scientific](https://m.media-amazon.com/images/I/61RX+vAb81L.jpg)
Amazon.com: Very High Level Hydrogen Peroxide H2O2 Test Strips, 2-10%, Time Based Test [Vial of 50 Strips] : Industrial & Scientific

SOLVED: 34 drinking water sample has a measured arsenic concentration of 1.28x10-= M: Convert the arsenic concentration to parts-per-million (ppm), assuming solution density of 1.00 g/mL: Is the water sample under the

Concentration of Hydrogen Peroxide in a 10 Volume Solution - An Interesting Stoichiometry Problem - YouTube

SOLVED: 2. Calculate the number density of CO2 (365 ppm) in the atmosphere at ground level (P = 984 hPa,T=+18 C) Consider the following chemical mechanism for the production of hydrogen peroxide (
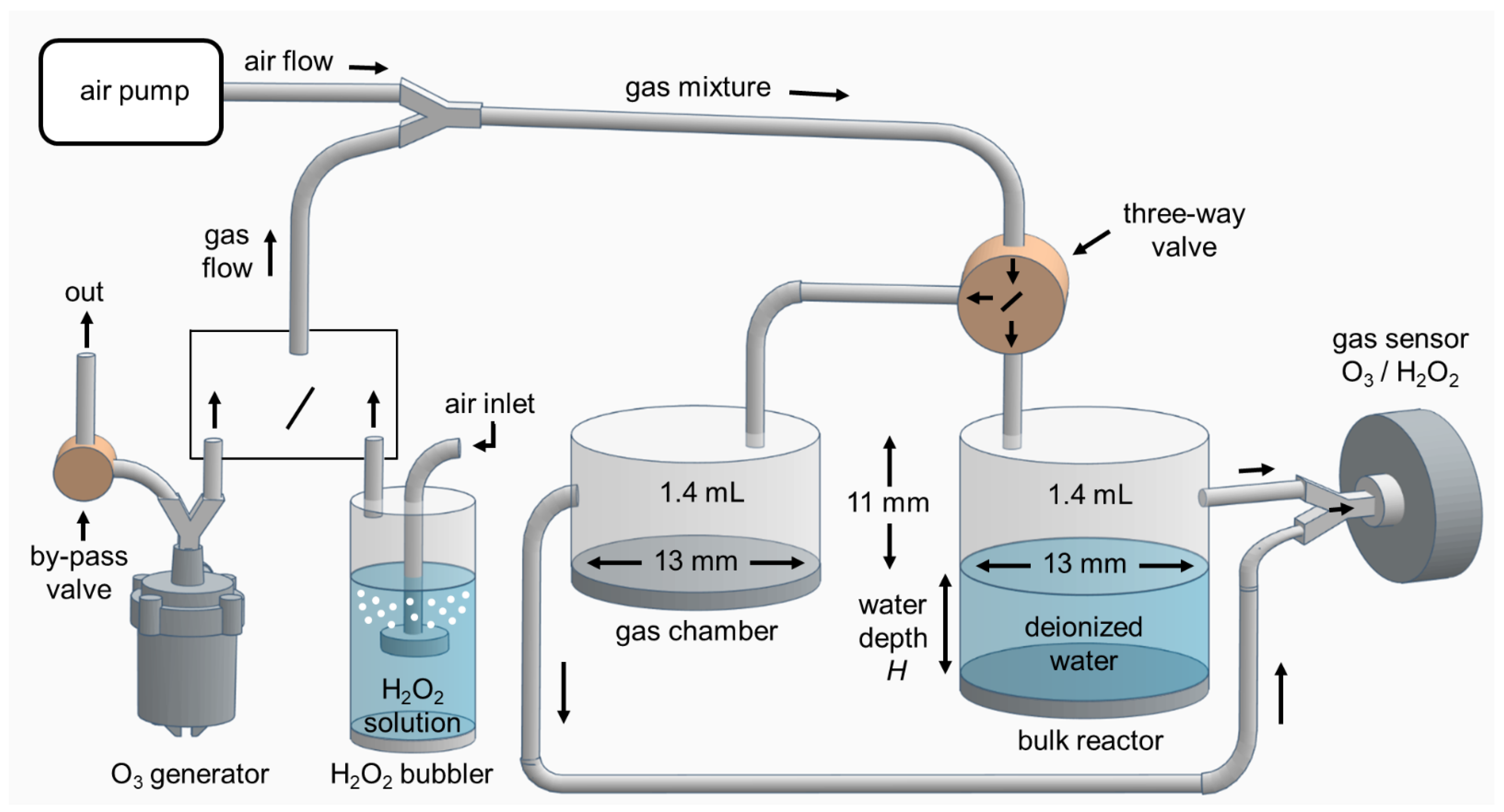
Water | Free Full-Text | Transport of Gaseous Hydrogen Peroxide and Ozone into Bulk Water vs. Electrosprayed Aerosol

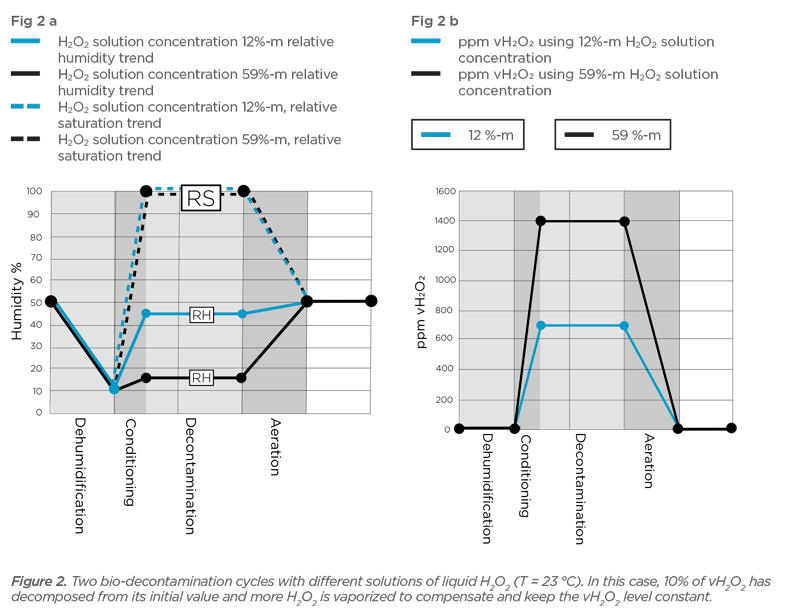
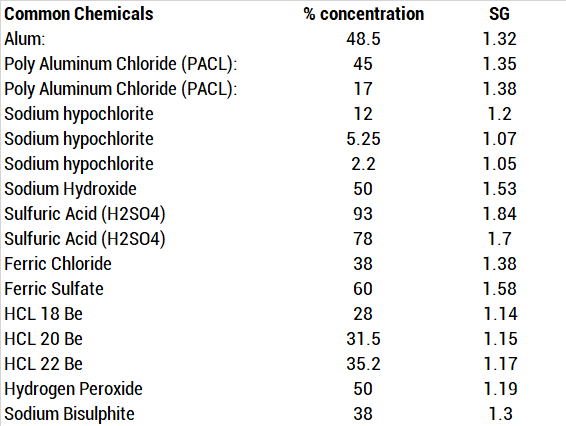
Impact of H2O2 Sorption by Polymers on the Duration of Aeration in Pharmaceutical Decontamination - Journal of Pharmaceutical Sciences

Quantification of photocatalytically-generated hydrogen peroxide in the presence of organic electron donors: Interference and reliability considerations - ScienceDirect