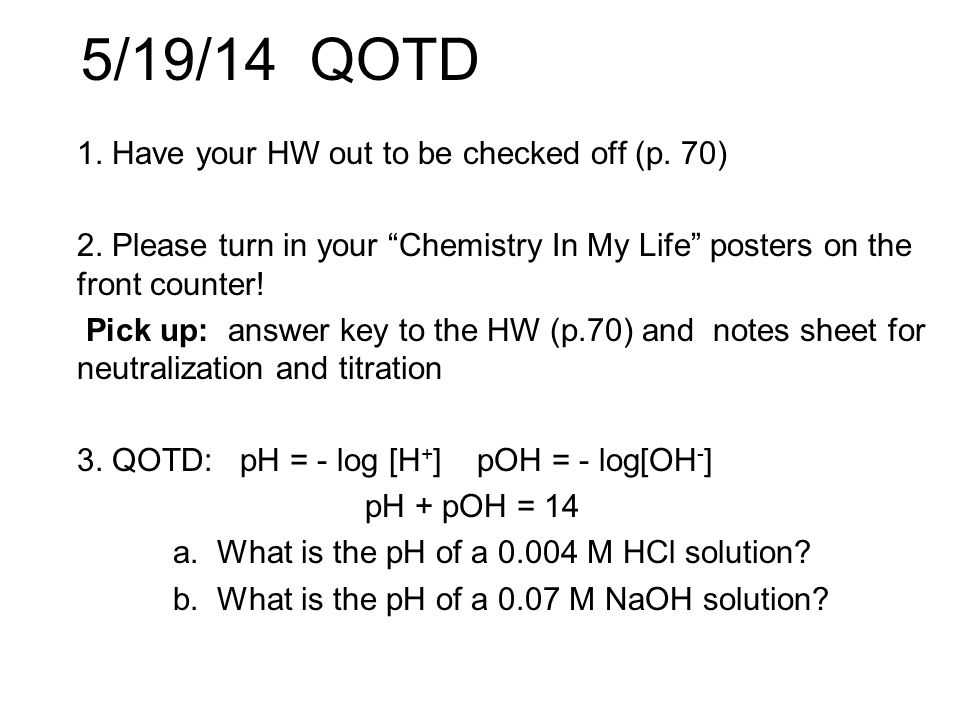
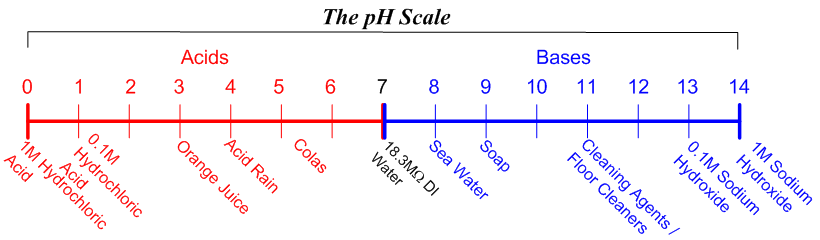
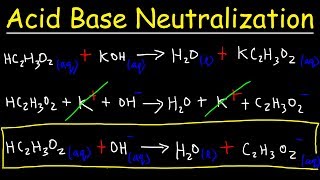
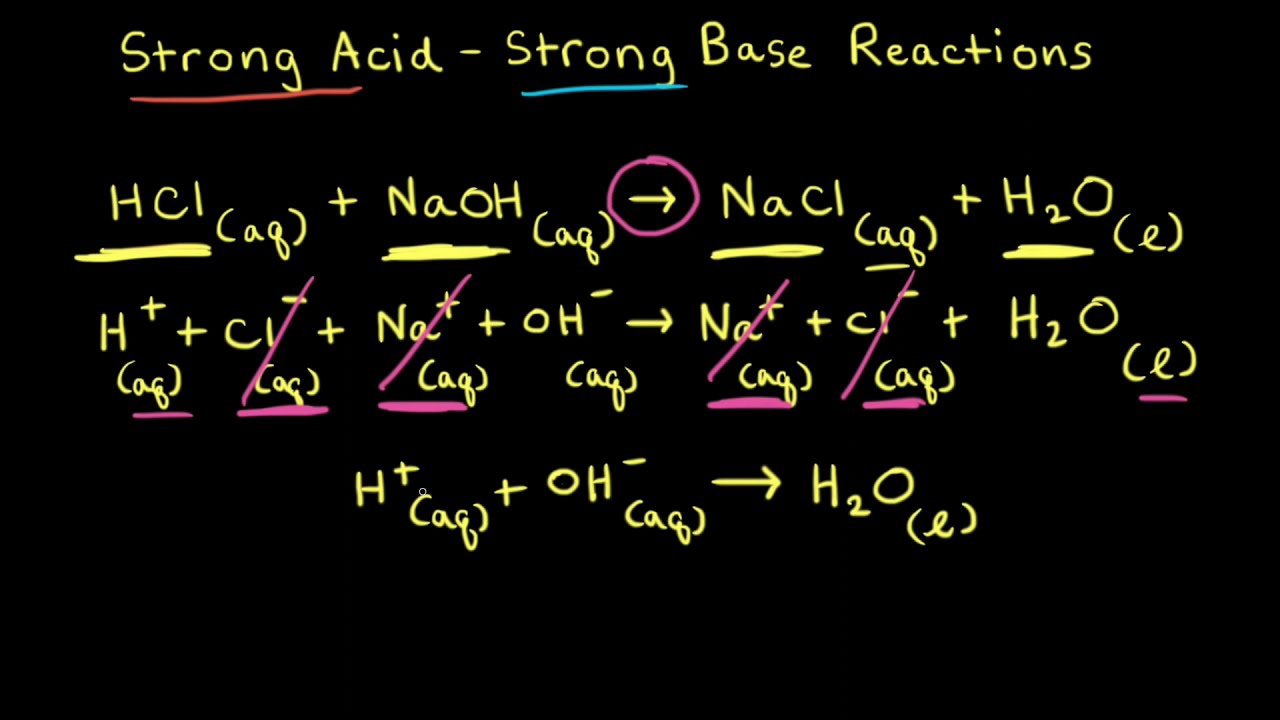SOLVED: Write a molecular equation for the neutralization reaction between aqueous HCI and aqueous Ca(OHJz? (3) The titration of 20.0-mL of a sulfuric acid solution of unknown concentration requires 22.87-mL of 0.158M
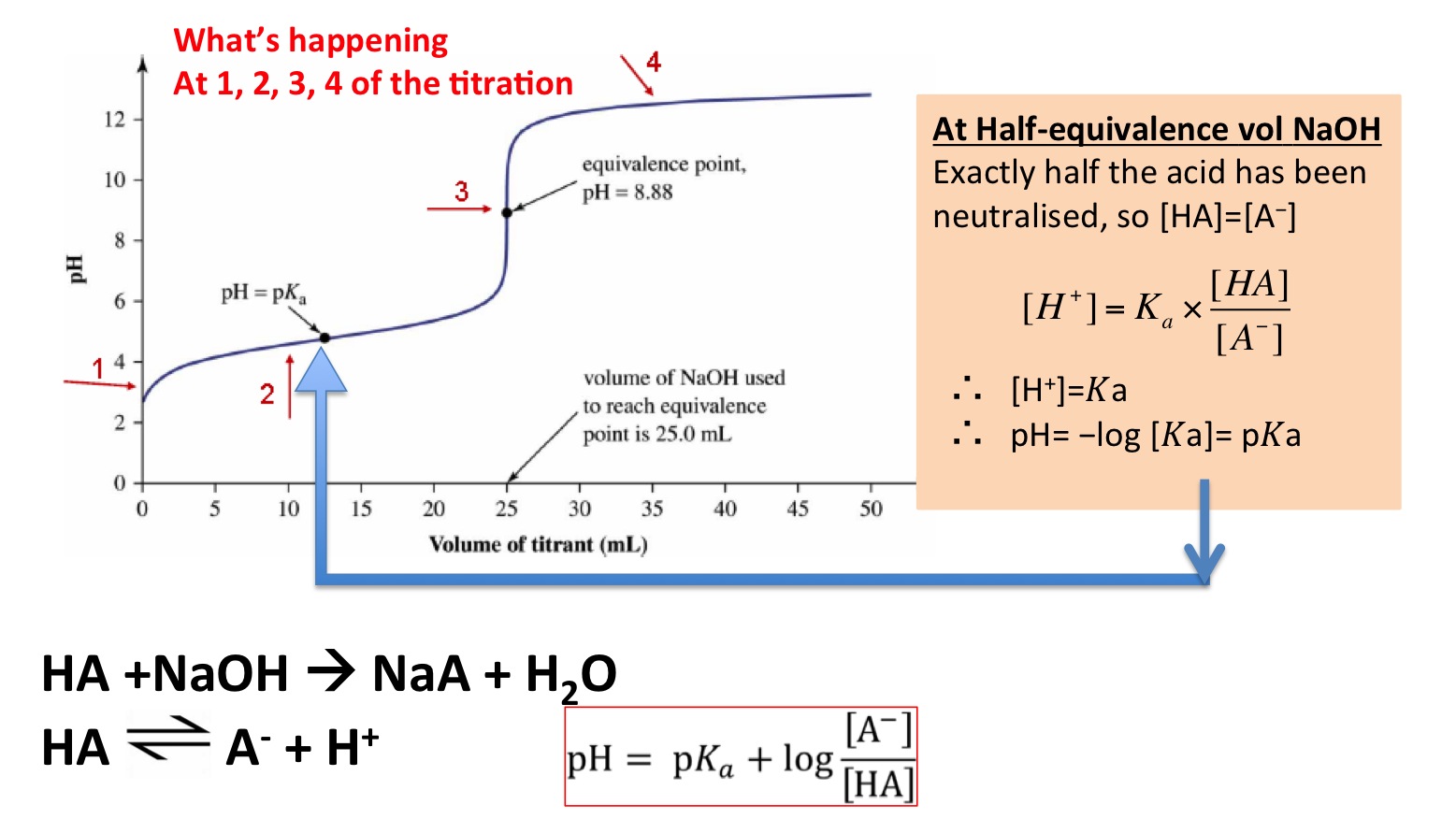
The "pH" at one-half the equivalence point in an acid-base titration was found to be 5.67. What is the value of K_a for this unknown acid? | Socratic

SOLVED: Practice Problem: Calculating pH during Titration Strong Base-Weak Acid ANSWER Calculate the pH of a solution formed when 50.0 mL of 0.100 M CH,COOH is titrated with 45.0 mL of 0.100
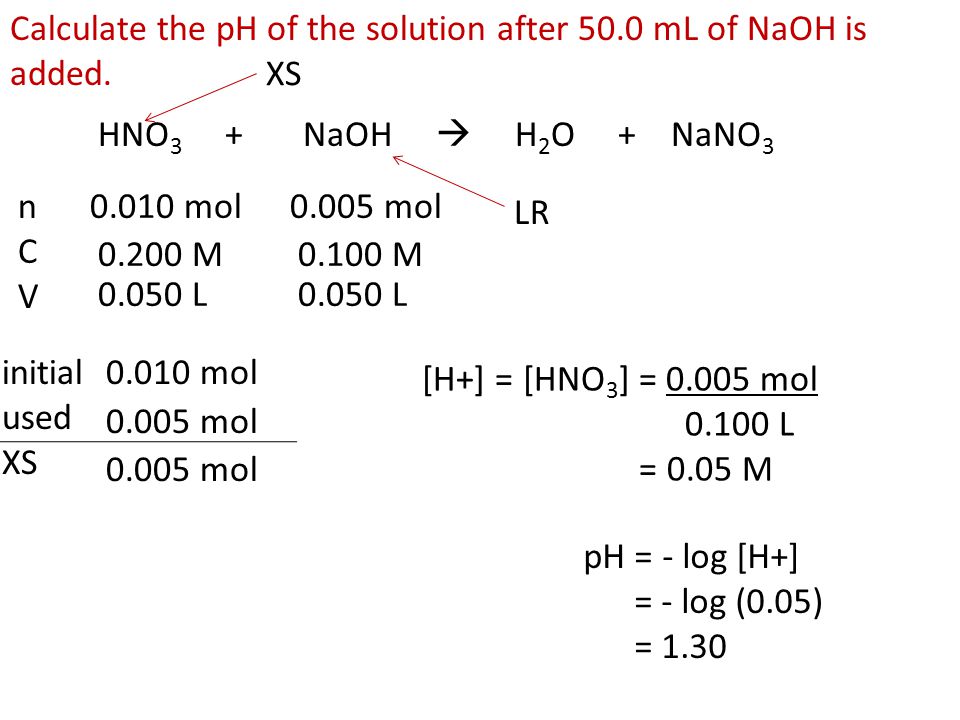
8.7 Acid-Base Titration Learning Goals … … determine the pH of the solution formed in a neutralization reaction. - ppt download